LAb 2a: dissecting a cell and examining its components
Purpose:
The purpose of this lab is to find out what positive and negative results fro, indicator tests for protein, carbohydrates, and fats. As well as, if parts of an egg test positive for protein, carbohydrate, and or fat.
Materials:
|
|
|
Procedure:
PART 1: Separating Cell Structures
PART 2: Testing Standard Solutions
Monosaccharide Indicator Standard Test:
Starch Indicator Standard Test
Protein Indicator Test:
Lipid Indicator Test:
PART 3: Molecular Composition of Egg Components
- Let 1 uncooked egg sit in white vinegar, covered by plastic wrap, for 24 to 48 hours. During which you will complete the second part of lab.
- After those 24-48 hours, gently pick up egg with slotted spoon and rinse with in water. What chemicals were involved as reactants and products?
- Feel the outside of the cell. The outside membrane being different from what is around the egg yolk provides a good model of a cell membrane. Place egg in beaker of 5% NaCI solution for another 24 hours. Describe what it looks like and what might have caused the change in appearance.
- Rinse off the egg with some more water and place in a beaker of distilled water for again another 24 hours. Describe how again the egg's appearance has changed and what might have changed it.
- Gently, slice open the egg's membrane and allow the egg white to drip through slotted spoon into a 100-mL beaker. Try not to pierce the yolk as you do this.
- Finally, place egg yolk on a separate beaker, and set aside the egg membranes.
PART 2: Testing Standard Solutions
Monosaccharide Indicator Standard Test:
- Mix 2 mL of a 2% glucose solution with 2 mL of a Benedict's solution. Heat for 2 minutes.
- Mix 2 mL of deionized water with 2 mL of Benedict's solution, heat for 2 minutes.
Starch Indicator Standard Test
- Mix 2 mL of starch suspension with 0.25 mL of Lugol's Iodine. Mix by gently swirling.
- Mix 2 mL of deionized water with 0.25 of Lugol's iodine. Gently swirl to mix.
Protein Indicator Test:
- Mix 2 mL of gelatin solution + 1 mL biuret solution and gently mix.
- Mix 2 mL of deionized water + 1 mL biuret solution and gently mix.
Lipid Indicator Test:
- Mix 60 uL Sudan IV solution to oil and gently mix.
- Mix 60 uL Sudan solution to deionized water and gently mix.
PART 3: Molecular Composition of Egg Components
- Finish all the Indicator tests
- Record results from testing the egg membranes
- Give each result a numerical value
Data and Results:
Part 1: Day to day checkups on our egg
Day 1:
We left the egg sitting in vinegar. We completely submerged the egg in vinegar, where we would leave it for the next 24 hours.
Day 2:
We removed the egg from vinegar and noticed the shell was dissolved. This might be due to vinegar's acidic nature that ate away at the eggs shell. We then left the egg sitting in a salt solution for the next 24 hours.
Day 3:
We checked on the egg again and the egg looked considerably smaller. This was most likely due to the egg sitting in salt water. The salt has moved to inside the egg, through its newly permeable shell while all the water has moved to outside the egg. This gives the egg a more shriveled look, similar to what your fingers look like after swimming in the ocean.
We rinsed the egg and left it in a beaker of de-ionized water for the next 24 hours.
Day 4:
The next day, we checked on the egg, which has increased in size overnight. This was most likely a result from soaking it in water. Since the egg's shell has been dissolved from the vinegar, the water is then allowed to pass through the now permeable membrane of the egg. All the water has been drawn inside the egg. We know this because when we later dissected it, the egg yolk, and white both burst out through the membrane when we applied slight pressure with a scalpel.
Part 2:
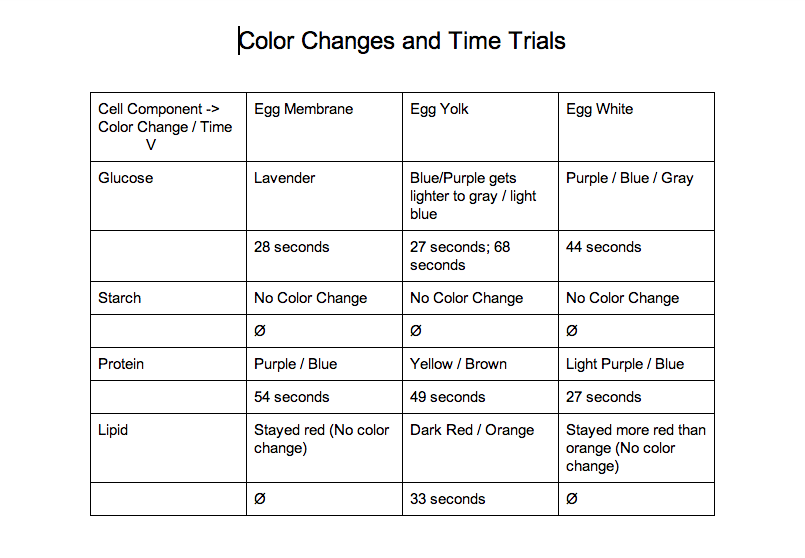
Here is a data table showing the color changes from each test:
Day 1:
We left the egg sitting in vinegar. We completely submerged the egg in vinegar, where we would leave it for the next 24 hours.
Day 2:
We removed the egg from vinegar and noticed the shell was dissolved. This might be due to vinegar's acidic nature that ate away at the eggs shell. We then left the egg sitting in a salt solution for the next 24 hours.
Day 3:
We checked on the egg again and the egg looked considerably smaller. This was most likely due to the egg sitting in salt water. The salt has moved to inside the egg, through its newly permeable shell while all the water has moved to outside the egg. This gives the egg a more shriveled look, similar to what your fingers look like after swimming in the ocean.
We rinsed the egg and left it in a beaker of de-ionized water for the next 24 hours.
Day 4:
The next day, we checked on the egg, which has increased in size overnight. This was most likely a result from soaking it in water. Since the egg's shell has been dissolved from the vinegar, the water is then allowed to pass through the now permeable membrane of the egg. All the water has been drawn inside the egg. We know this because when we later dissected it, the egg yolk, and white both burst out through the membrane when we applied slight pressure with a scalpel.
Part 2:
Here is a data table showing the color changes from each test:
Part 3:
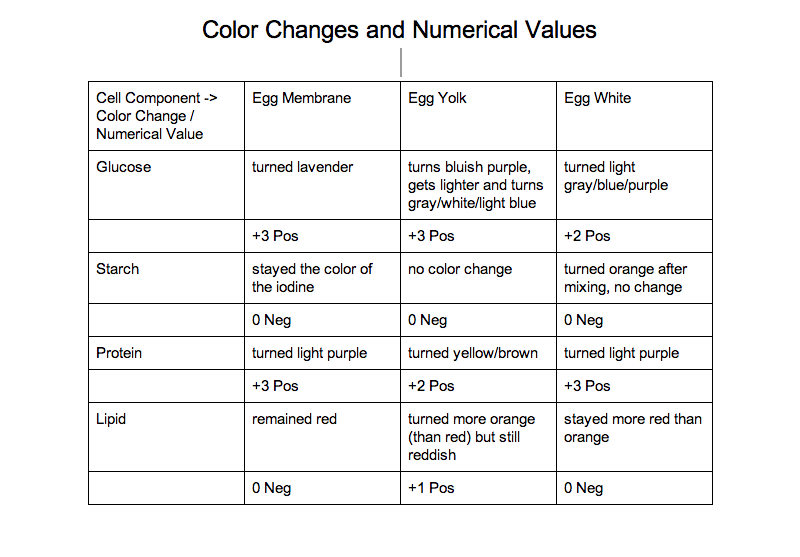
Below is a chart of all the color changes and a numerical value explaining the strength of the result:
Below is a chart of all the color changes and a numerical value explaining the strength of the result:
Reflection:
|
I enjoyed many components of this lab. I found it really interesting when testing negative and positive indicator tests. It was fun to mix together different components and see the reaction. For example, testing for glucose, it was cool to see how the two substances reacted together over heat. It was even more interesting to then use what we learned from completing the indicator tests to apply it to testing the different egg components for glucose, starch, protein, and lipids.
|
Me and my partner, Bronte, collaborated very well. We finished all the experiments with time to spare. Together, we efficiently each did parts of the lab and prepared the next part while the other was working. Altogether, we worked together very well and learned a lot from this lab.
|
If I could do this lab all over again, I would do some things differently. Firstly, I would probably divide up the work. While we worked rather efficiently, we still could have divided up the lab work even more. For example, one of us could measure out and combine the substances for one test while the other prepares another test, and then afterwards we can collaborate and discuss the changes we observed.
|
Conclusion: analyzing the data
Through our data we have learned a lot about what an egg contains. After completing our tests we found out that there is sugar and protein in an egg's shell. We also learned that there are sugars, protein, and lipids in an egg yolk. Finally, we learned that there is sugar and protein in egg whites.
While overall our experiment was a success, we still had some errors. This could be due to many factors. For example, when using the "hot water bath" the burners take a while to heat up and may not have been at the right temperature when they were used.
Another way we could apply the indicator tests is in medicine. For example testing for diabetes, you test your pee for glucose.
While overall our experiment was a success, we still had some errors. This could be due to many factors. For example, when using the "hot water bath" the burners take a while to heat up and may not have been at the right temperature when they were used.
Another way we could apply the indicator tests is in medicine. For example testing for diabetes, you test your pee for glucose.